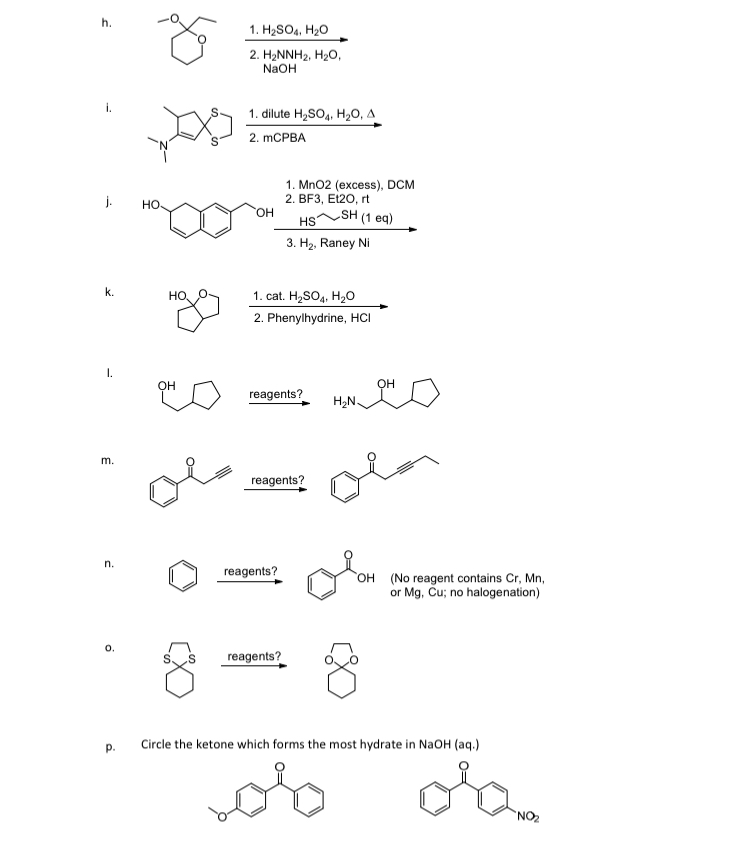
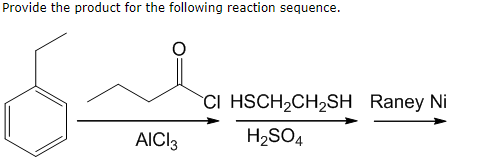
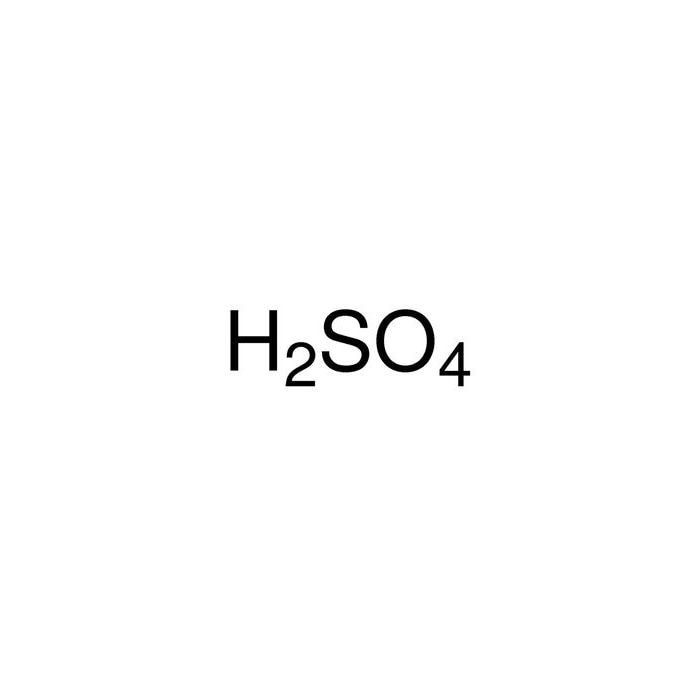
![4] Complete and balance the following organic reactions (ii) to (v) and match the names of each reaction from list (2) to (5). One has been done you as an example. Conc. 4] Complete and balance the following organic reactions (ii) to (v) and match the names of each reaction from list (2) to (5). One has been done you as an example. Conc.](https://toppr-doubts-media.s3.amazonaws.com/images/4903703/da785a60-cf16-4f6b-8427-bb073f7a8246.jpg)
4] Complete and balance the following organic reactions (ii) to (v) and match the names of each reaction from list (2) to (5). One has been done you as an example. Conc.

Processes | Free Full-Text | Incidence of Electric Field and Sulfuric Acid Concentration in Electrokinetic Remediation of Cobalt, Copper, and Nickel in Fresh Copper Mine Tailings
Effect of H2SO4 concentration on Ni leaching. Conditions: (a) 6 vol.%... | Download Scientific Diagram

Ni+H2SO4=H2+Ni2(SO4)3 Balanced Equation||Nickel+Sulphuric acid=Hydrogen+Nickel sulphate Balanced Equ - YouTube

Kinetics of nickel leaching from low-nickel matte in sulfuric acid solution under atmospheric pressure - ScienceDirect

A Little Nickel Goes a Long Way: Ni Incorporation into Rh2P for Stable Bifunctional Electrocatalytic Water Splitting in Acidic Media | ACS Materials Au